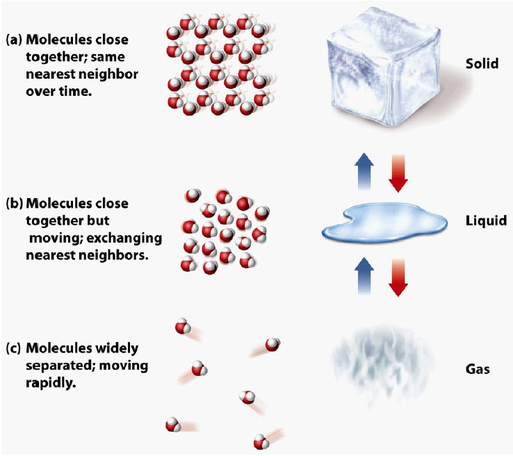
Liquids and Solids
The three states of matter are solid, liquid, and gas.
Solid
|
Liquid
|
Gas
|
Evaporation
|
Condensation
|
Fog and Clouds
|
Boiling
|
Heat Equations
Heat of Fusion:
- The amount of energy/mol needed to convert between solid and liquid.
- H2O ~ 6 kJ/mol
- The amount of energy/mol needed to convert between liquid and gas.
- H2O ~ 41kJ/mol
Forces
- Intramolecular Forces: forces in a molecule
- Ex. covalent bonds, ionic bonds
- Intermolecular Forces: forces between molecules
- Ex. dipole-dipole forces: attraction between polar molecules; stronger than dispersion forces
- Ex. hydrogen bonding: attraction b/t partial positive and neg.
- Ex. london dispersion forces
- Hydrogen bonds are the strongest type of intermolecular forces between different molecules, BUT ionic and covalent bonds (holding ions or atoms together in compounds) are stronger than hydrogen bonds!
Vapor Pressure
- In a sealed container, a given amount of liquid in a container will decrease slightly.
- Eventually, the volume will be come constant.
- The rate of evaporation and condensation will equal.
sublimation: the process of changing from a solid to a gas without passing through an intermediate liquid phase
deposition: the process of changing from a gas to a solid without passing through an intermediate liquid phase
deposition: the process of changing from a gas to a solid without passing through an intermediate liquid phase
Crystalline SolidsIonic Solid: components are ions
|
ionic solids:
|
Alloys
- A substance that contains a mixture of elements and has metallic properties.
- Substitutional Alloy: substitutes an element in a molecule
- Ex. brass
- Interstitial Alloy: adds an element in between the molecules
- Ex. Steel