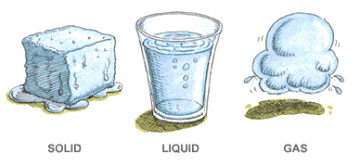
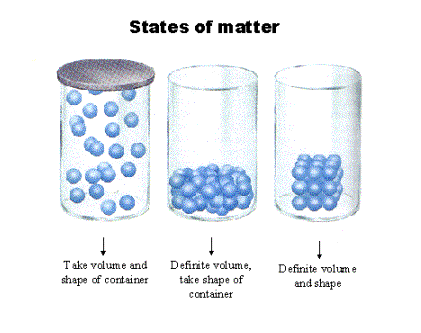
Section 1: Matter
- Matter has 2 characteristics: mass and it occupies space.
|
|
Section 2: Physical and Chemical Properties and Changes
|
Examples of physical properties:
Examples of chemical properties:
Example of physical changes:
Example of chemical changes:
|
|
Section 3: Elements and Compounds
|
Examples of elements:
Examples of compounds:
|
|
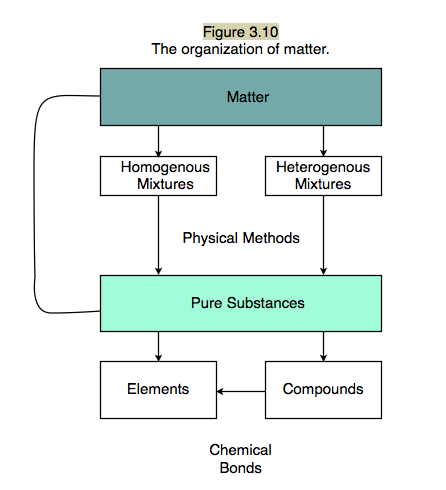
Section 4: Mixtures and Pure Substances
Examples of mixtures:
- wood
- wine
- coffee
- water pumped from deep in the earth
- Most of matter consists of mixtures of substances.
- Air is a complex mixture of gases.
- Pure substances are either elements or compounds.
- Pure water always has the same physical and chemical properties and is always made of molecules containing hydrogen and oxygen in exactly the same proportions, regardless of original source of the water.
- Mixtures can be separated into pure substances: elements and/or compounds.
- Example: Mixture known as air can be separated into oxygen (element), nitrogen (element), water (compound), carbon dioxide (compound), argon (element), and other pure substances.
- Example of a pure substance: copper metal
- Example of a homogenous mixture: gasoline, air, brass
- Example of a heterogenous mixture: a stream with a gravel at the bottom