Section 1: Naming Compounds
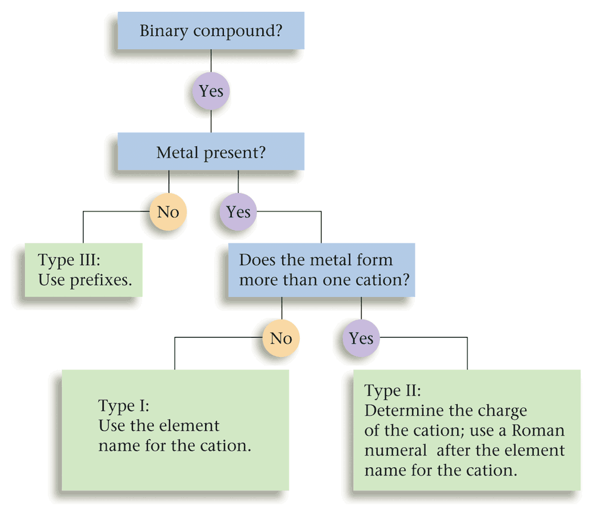
- We can divide binary compounds into two broad classes:
- Compounds that contain a metal and a nonmetal.
- Compounds that contain two nonmetals.
Section 2: Naming Binary Compounds That Contain a Metal and a Nonmetal (Types I and II)
|
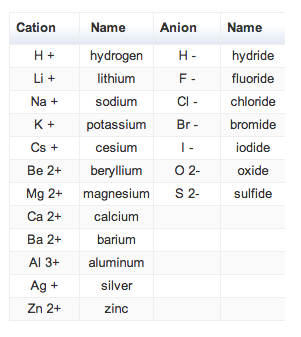
Common Simple Cations and Anions |
|
Rules for Naming Type I Ionic Compounds:
|
Examples:
|
|
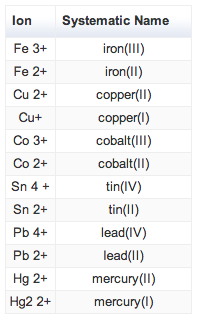
Rules for Naming Type II Ionic Compounds:
|
Examples:
|
Common Type II Cations |
Section 3: Naming Binary Compounds That Contain Only Nonmetals (Type III)
|
Rules for Naming Type III Binary Compounds:
|
Examples:
|
Section 4: Naming Binary Compounds: A Review
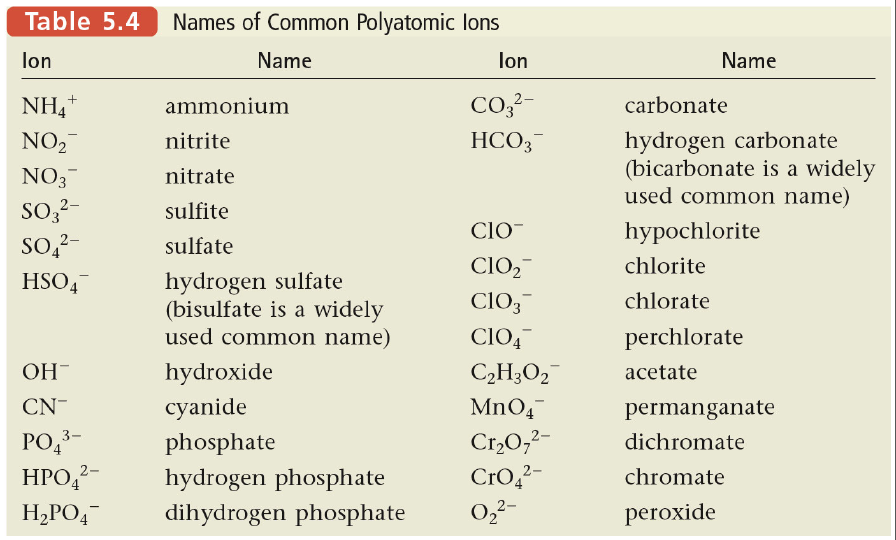
Section 5: Naming Compounds That Contain Polyatomic Ions
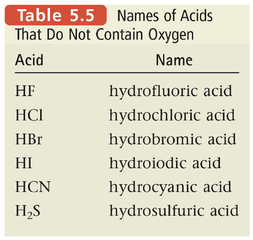
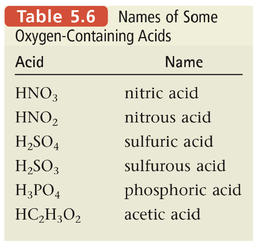
Section 6: Naming Acids
|
Section 7: Writing Formulas from Names
- Writing the formula involves using all of the information that you have learned from this chapter.
- Example Problems: Give the formula for each of the following compounds.
- Potassium Hydroxide KOH
- Sodium Carbonate Na2CO3
- Nitric Acid HNO3
- Cobalt(III) Nitrate Co(NO3)3
- Calcium Chloride CaCl2
- Lead(IV) Oxide PbO2
- Dinitrogen Pentoxide N2O5
- Ammonium Perchlorate NH4ClO4
- Vanadium(V) Fluoride VF5