Measurements and Calculations
Major Points/Concepts
- measurement: a quantitative observation
- A measurement must always consist of a number and a unit.
- Example: 125 can be written as the product of 125 = 1.25 * 100
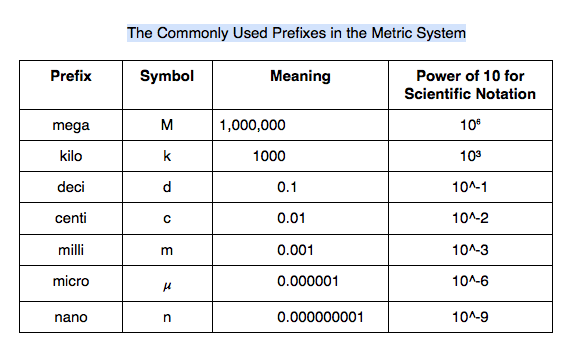
- scientific notation: simply expresses a number as a product of a number between 1 and 10 and the appropriate power of 10
- Example: 75,000 = 7.5 * 10,000 = 7.5 * 10⁴
- How to determine the appropriate power of 10.
⤾ ⤾ ⤾ ⤾
- Because the exponent is positive, it moved to the left.
- Example: 0.000038 = 3.8 * 10^-5
- How to determine the appropriate power of 10.
⤿ ⤿ ⤿ ⤿
- Because the exponent is negative, it moved to the right.
- HINT: Remember that Left Is Positive; LIP.
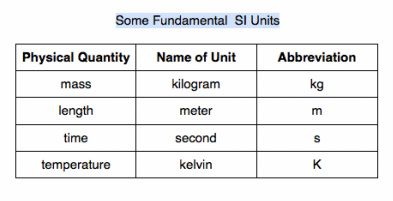
- units: part of a measurement that tells us what scale or standard is being used to represent the results of the measurement
- English system: system that is used in the United States
- metric system: system used in most of the rest of the industrialized world
- International System (SI): are based on the metric system and units derived from the metric system
- Measurements consist of both a number and a unit, and both are crucial.
|
- It is very important to realize that a measurement always has some degree of uncertainty. The uncertainty of a measurement depends on the measuring device.
- significant figures: numbers recorded in a measurement (all the certain numbers plus the first uncertain number)
- Rules for Counting Significant Figures:
- Nonzero Integers: always count as significant figures
- Zeros: There are three class of zeros.
- Leading zeros that precede all of the nonzero digits. They never count as significant figures. (Example: 0.0029, The number only has 2 significant figures, the two and the nine.)
- Captive zeros that fall between nonzero digits. They always count as significant figures. (Example: 100827, The number has 6 significant figures.)
- Trailing zeros are zeros at the right end of the number. They are only significant if the number has a decimal. (Example: 100, The number has 1 significant figure. 0.040, The number has 4 significant figures.)
obtained using measuring devices but were determined by
counting, like 10 or 28
- Rounding Off Numbers:
- rounding off: reduce it to fewer digits
- Example: 6.3852 to 6.4
- Determining Significant Figures in Calculations:
- For multiplication or division, the number of significant figures in the result is the same as that in the measurement with the smallest number of significant figures. This measurement is limiting, because it limits the number of significant figures in the result.
- Example: 2.24 * 1.7 = 3.808 to 3.8
- Example: 8.392 / 284 = 0.0295492... to 2.95 * 10^-2
- For addition or subtraction, the limiting term is the one with the smallest number of decimal places.
- Example: 12.11 + 18.0 + 1.013 = 31.123 to 31.1
- Example: 0.6875 - 0.1 = 0.5875 to 0.6
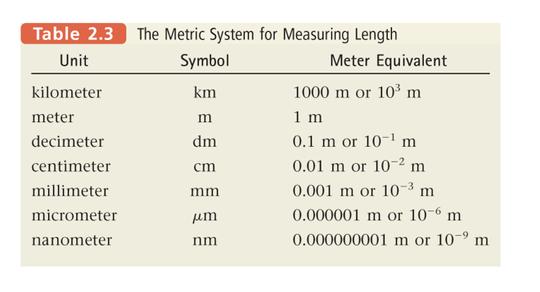
- Convert from One Unit to Another:
- To convert from one unit to another, use the equivalence statement that relates the two units. The conversion factor needed is a ratio of the two parts of the equivalence statement.
- Choose the appropriate conversion factor by looking at the direction of the required change (make sure the unwanted units cancel).
- Multiply the quantity to be converted by the conversion factor to give the quantity with the desired units.
- Check that you have the correct number of significant figures.
- Ask whether your answer makes sense.
- density: amount of matter present in a given volume of a substance
- Density = mass/volume
- specific gravity: ratio of the density of a given liquid to the density of water at 4 degrees Celsius
Online Resources
- http://chemistry.about.com/od/mathsciencefundamentals/a/sigfigures.htm
Video Resources
- http://www.youtube.com/watch?v=REtBibhIqfo
- http://www.youtube.com/watch?v=H0vFB129gd4
Activity Resource
- http://quizlet.com/3150800/chemistry-unit-3metric-units-flash-cards/