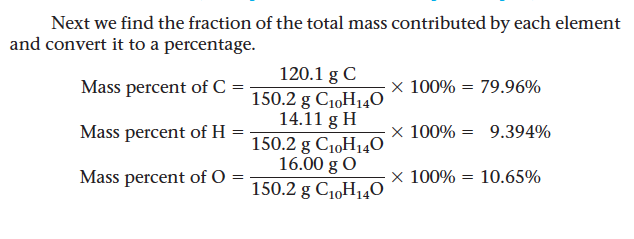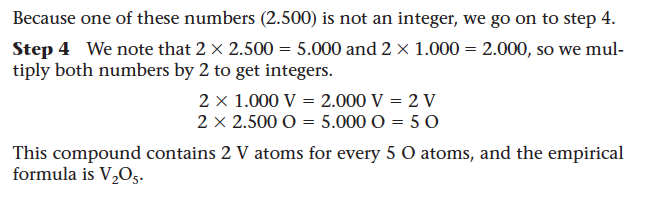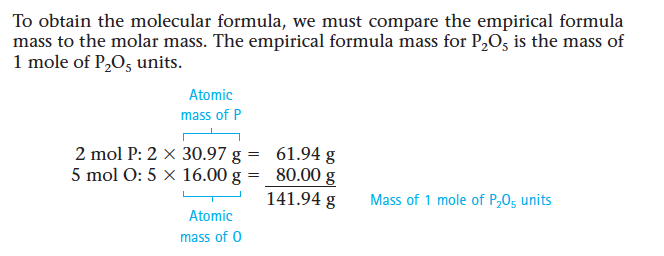Chemical Composition
Section 1: Counting by Weighing
Section 2: Atomic Masses: Counting Atoms by Weighing
|
Section 3: The Mole
- Samples in which the ratio of the masses is the same as the ratio of the masses of the individual atoms always contain the same number of atoms.
- The mole (abbreviated mol) can be defined as the number equal to the number of carbon atoms in 12.01 grams of carbon. Techniques for counting atoms very precisely have been used to determine this number to be 6.022 * 10^23 . This number is called Avogadro’s number. One mole of something consists of 6.022 * 10^23 units of that substance. Just as a dozen eggs is 12 eggs, a mole of eggs is 6.022 * 10^23 eggs.
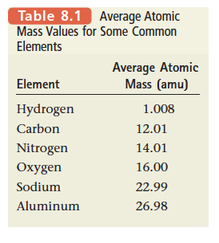
- A sample of an element with a mass equal to that element’s average atomic mass expressed in grams contains 1 mole of atoms.
Section 4: Learning To Solve Problems
- It is our purpose in this text to help you learn to solve problems in a flexible, creative way based on understanding the fundamental ideas of chemistry. We call this approach conceptual problem.
Section 5: Molar Mass
- A chemical compound is a collection of atoms.
- Remember that the least number of decimal places limits the number of significant figures in addition.
- Ex. The formula for sulfur dioxide is SO2 , which means that 1 mole of SO2 molecules contains 1 mole of sulfur atoms and 2 moles of oxygen atoms.
- We need to find the mass of 1 mole of SO2 molecules, which is the molar mass of SO2 .
mass of 2 mol O = 2* 16.00 = 32.00 g
mass of 1 mol SO2 = 64.07 g (molar mass)
Section 6: Percent Composition of Compounds
- The mass fraction is converted to mass percent by multiplying by 100%.
- Ex. Carvone is a substance that occurs in two forms, both of which have the same molecular formula (C10 H14 O) and molar mass. One type of carvone gives caraway seeds their characteristic smell; the other is responsible for the smell of spearmint oil. Compute the mass percent of each element in carvone.
Section 7: Formulas of Compounds
- When we break a compound down into its separate elements and “count” the atoms present, we learn only the ratio of atoms—we get only the relative numbers of atoms.
- The formula of a compound that expresses the smallest whole-number ratio of the atoms present is called the empirical formula or simplest formula.
- The actual formula of a compound—the one that gives the composition of the molecules that are present—is called the molecular formula.
- Ex. C6H6. This is the molecular formula for benzene, a liquid commonly used in industry as a starting material for many important products.
- C6H6 (CH)6; CH is the empirical formula. Each subscript in the empirical formula is multiplied by 6 to obtain the molecular formula.
Section 8: Calculation of Empirical Formulas
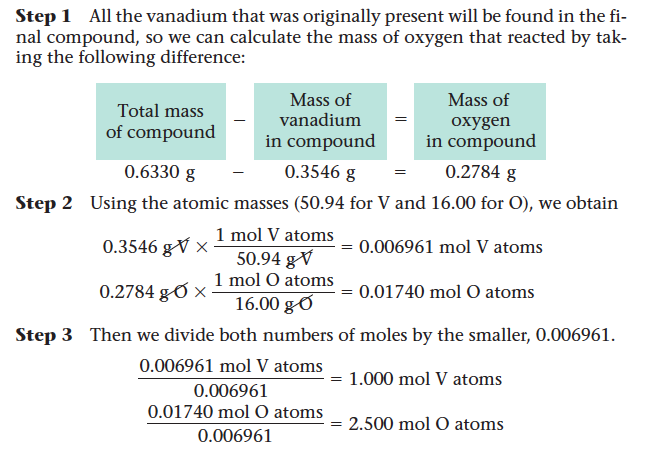
- Step 1: Obtain the mass of each element present (in grams).
- Step 2: Determine the number of moles of each type of atom present.
- Step 3: Divide the number of moles of each element by the smallest number of moles to convert the smallest number to 1. If all of the numbers so obtained are integers (whole numbers), these are the subscripts in the empirical formula. If one or more of these numbers are not integers, go on to step 4.
- Step 4: Multiply the numbers you derived in step 3 by the smallest integer that will convert all of them to whole numbers. This set of whole numbers represents the subscripts in the empirical formula.
- Ex. When a 0.3546-g sample of vanadium metal is heated in air, it reacts with oxygen to achieve a final mass of 0.6330 g. Calculate the empirical formula of this vanadium oxide.
- We want to determine the empirical formula for the vanadium oxide, VxOy . That is, we want to solve for x and y .
- The compound contains 0.3546 g of vanadium and has a total mass of 0.6330 g. We know the atomic masses of vanadium (50.94 g/mol) and oxygen (16.00 g/mol).
Section 9: Calculation of Molecular Formulas
- If we know the composition of a compound in terms of the masses (or mass percentages) of the elements present, we can calculate the empirical formula but not the molecular formula.
- Ex. A white powder is analyzed and found to have an empirical formula of P2O5. The compound has a molar mass of 283.88 g. What is the compound’s molecular formula?