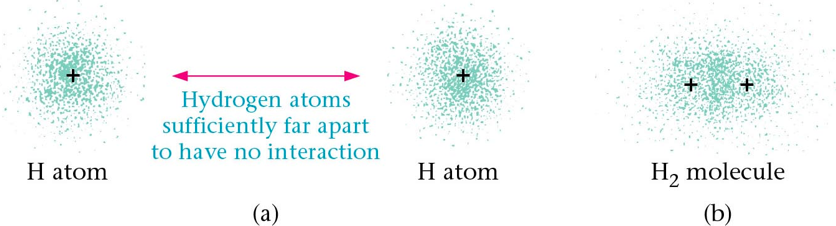
Section 1: Types of Chemical Bonds
|
|
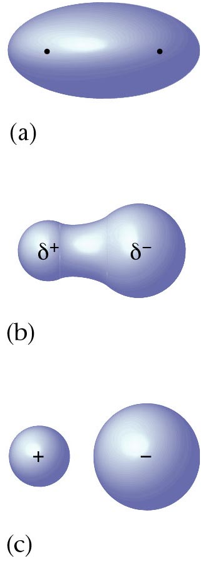
- The most logical explanation for the development of bond polarity (partial positive and negative charges on the atoms in molecules) is that the electrons in the bonds are not shared equally.
Section 2: Electronegativity
- When differ nonmetals react, a bond forms in which electrons are shared unequally, giving a polar covalent bond.
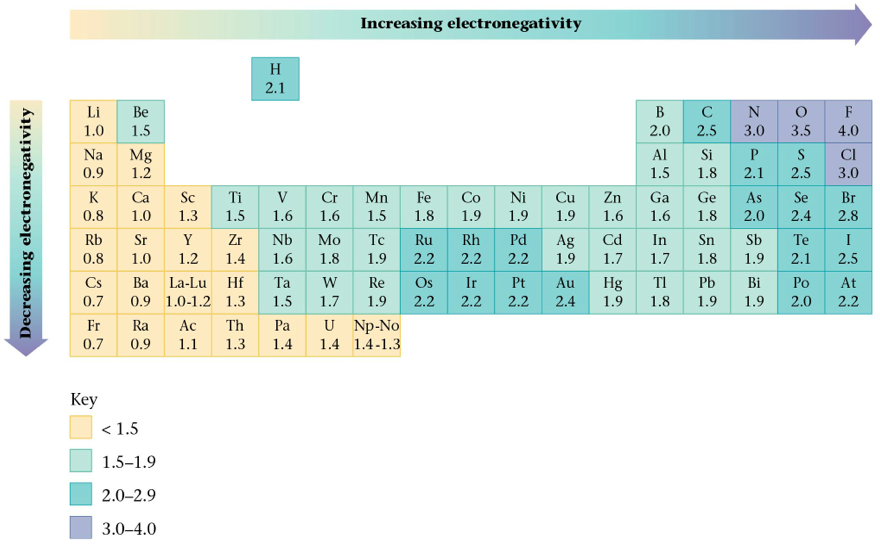
- Chemists determine electronegativity values for the elements by measuring the polarities of the bonds between differ atoms.
- Remember: The higher the atom's electronegativity value, the closer the shared electrons tend to be to that atom when it forms a bond.
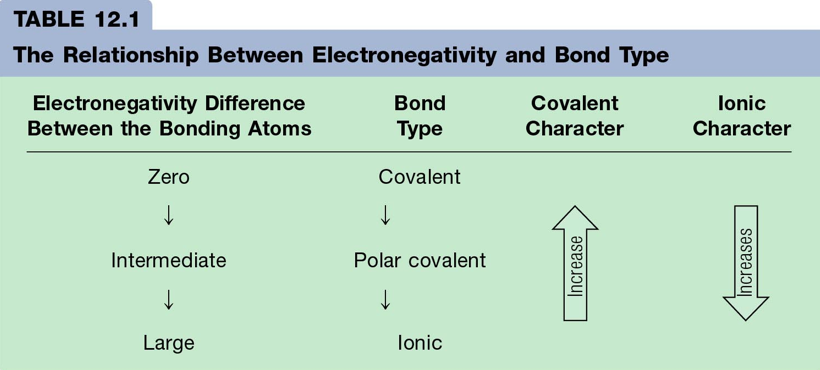
- The polarity of a bond depends on the difference between the electronegativity values of the atoms forming the bond.
- larger difference = more polar
- smaller difference = more equally shared
Section 3: Bond Polarity and Dipole Moments
- Any diatomic (two-atom) molecule that has a polar bond has a dipole moment.
- Some poly atomic molecules have dipole moments.
- The dipolar character of a molecule is often represented by an arrow. The arrow points towards the negative charge center and its tail indicates the positive center of charges.
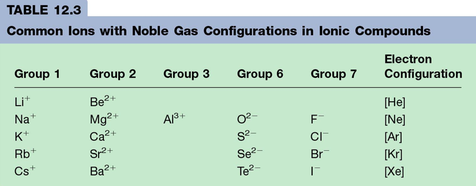
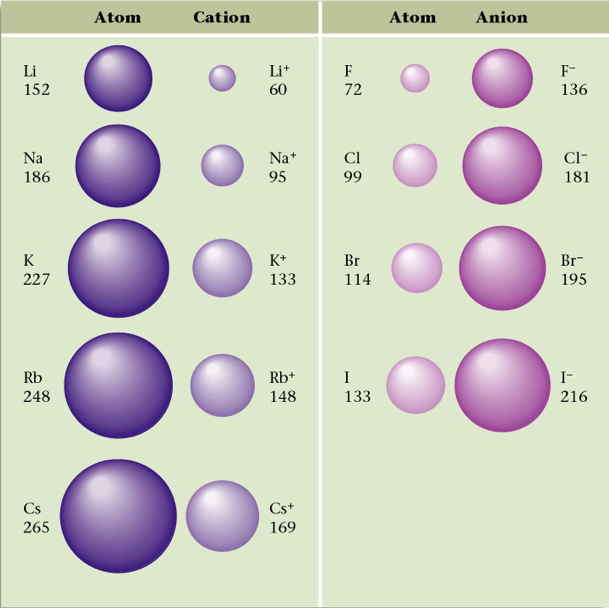
Section 4: Stable Electron Configurations and Charges on Ions
|
|
Section 5: Ionic Bonding and Structures of Ionic Compounds
|
Section 6: Lewis Structures
|
Steps for Writing Lewis Structures:
|
Section 7: Lewis Structures of Molecules with Multiple Bonds
|
Section 8: Molecular Structure
|
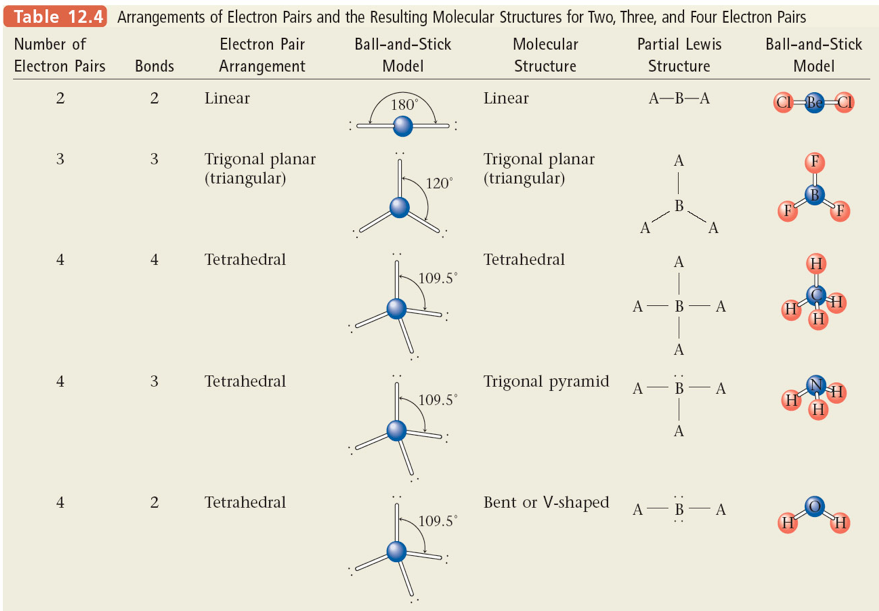
Section 9: Molecular Structure: The VSEPR Model
Steps for Predicting Molecular Structure Using the VSEPR Model
|
|