Section 1: Rutherford's Atom
|
|
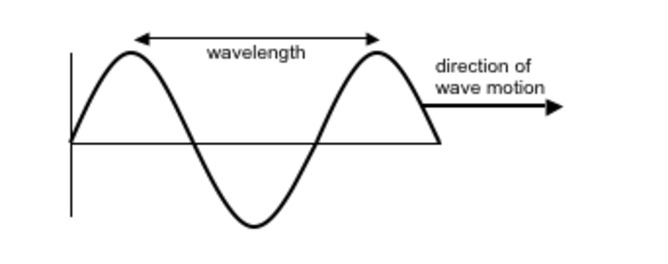
Section 2: Electromagnetic Radiation
|
Section 3: Emission of Energy by Atoms
- When atoms receive energy from some source - they become excited - they can release this energy by emitting light.
- The emitted energy is carried away by a photon.
- The energy of the photon corresponds exactly to the energy change experienced by the emitting atom.
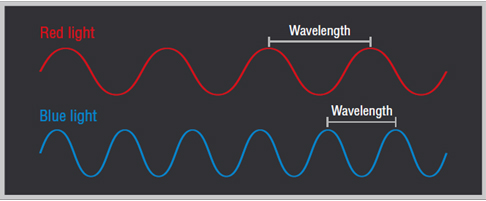
- High-energy protons correspond to short-wavelength light.
- Low-energy photons correspond to long-wavelength light.
Section 4: The Energy Levels of Hydrogen
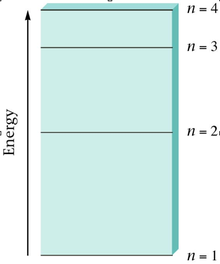
- Electrons can be found in the ground state. When energy is added to the atom electron can jump to higher energy levels. The electrons are considered in the excited state. These electron can relax down to a lower energy level and release the energy as light.
- Different wavelengths of light carry different amounts of energy per photon.
- Early on, scientist believed that atoms can exist on any energy levels.
- Because only certain photons are emitted, we know that only certain energy changes are occurring.
- There are certain discrete energy level that will always emit photons.
- There are quantized energy levels vs. continuous energy levels.
Section 5: The Bohr Model of the Atom
Section 6: The Wave Mechanical Model of the Atom
Section 7: The Hydrogen Orbitals
|
|
|
Section 8: The Wave Mechanical Model: Further Development
- Bohr model was discarded because it did not apply to all atoms.
- The wave mechanical model fits well with the design of the periodic table.
- Two electrons of opposite spin occupy each orbital. Represented by two arrows in opposite directions. ↑↓
- Principal Components of the Wave Mechanical Model of the Atom:
2. Energy of the level increases as the value of n increases.
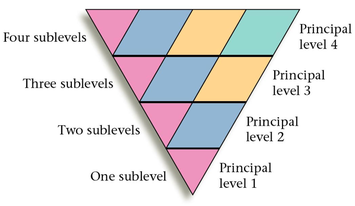
3. Each principal energy level contains one or more types of orbitals or sublevels.
4. Number of sublevels in a principal energy level equals n.
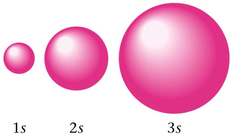
- PEL=1, it has 1 sublevel (1s)
- PEL=2, it has 2 sublevels (2s and 2p)
- PEL=3, it has 3 sublevels (3s, 3p, and 3d)
- PEL=4, it has 4 sublevels (4s, 4p, 4d, and 4f)
6. An orbital can be empty or have 1 or 2 electrons, but never more than 2. If 2 electrons occupy the same orbital, they must have opposite spins.
7. Shape of an orbital does not indicate the movement of the electrons. It indicates the probability distribution for an electron residing in that orbital.
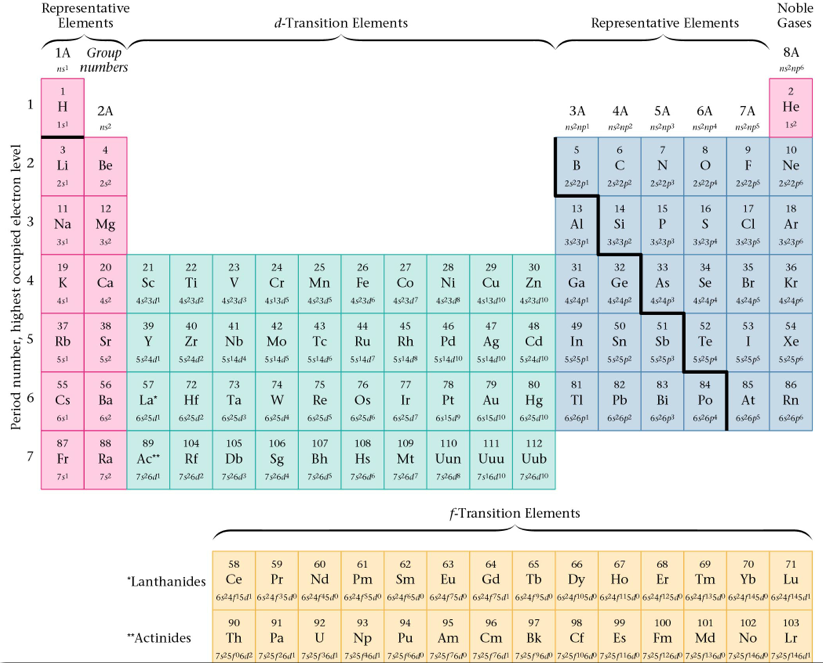
Section 9: Electron Arrangements in the First Eighteen Atoms on the Periodic Table
- valence electrons: the electrons in the outermost (highest) principal energy level of an atom
- Example: Nitrogen has an electron configuration of 1s22s22p3. It has electrons in PELs 1 and 2. Level 2 is your valence level and the electrons in 2s and 2p are your valence electrons.
- Only valence electrons are involved in bonding so they are the most important to chemists.
- Core electrons are in the innermost (lowest) principal energy levels.
- The atoms of elements in the same group(vertical column) have the same number of electrons in a given type of orbital (sublevel). The orbitals will be in different energy levels. Attributes to the fact that they have similar properties.
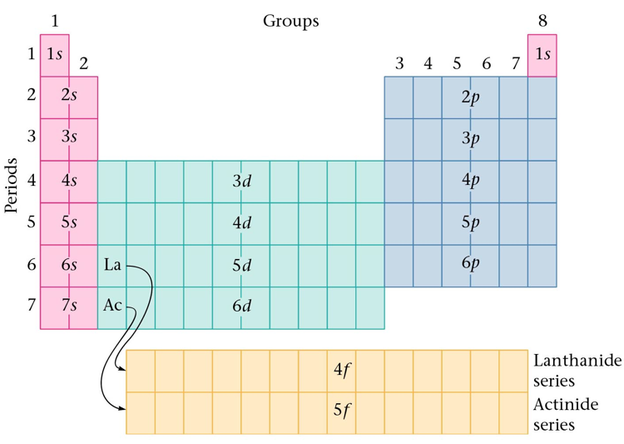
Section 10: Electron Configurations and the Periodic Table
- Orbital Filling:
- In a PEL that has d orbitals, the s orbital from the next fills before the d orbitals in the current level.
- After lanthanum, a group of fourteen elements occur.
- After actinium, a group of fourteen elements occur.
- Except for helium, the group numbers show the sum of electrons in the ns and np orbitals in the highest principle energy level that contains electrons.
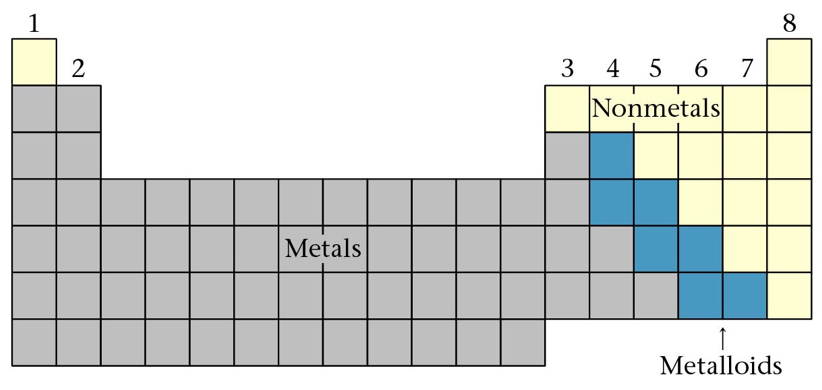
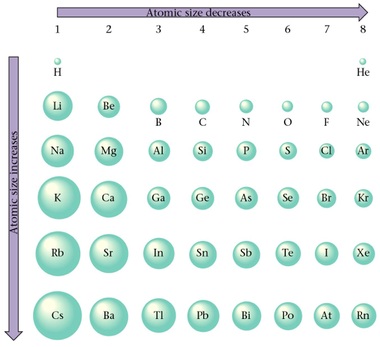
Section 11: Atomic Properties and the Periodic Table
|
|