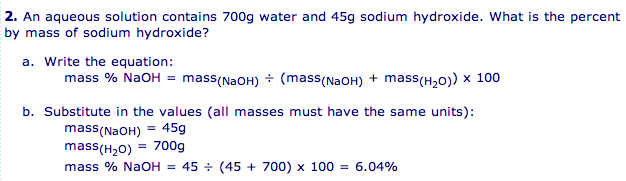Solution
- Solution is a homogenous mixture.
- A mixture in which the compounds are uniformly intermingled
- aka they are mixed well so that you can’t see the parts
- Two parts of a solution:
- Solvent: substance that present in the largest amount
- Solute: substance present in a small amount when compared a substance that is present in a large amount
- Ionic compound will dissolve in water since ionic compounds are made up of cation(+) and anion(-).
- Water will also dissolve many nonionic compounds.
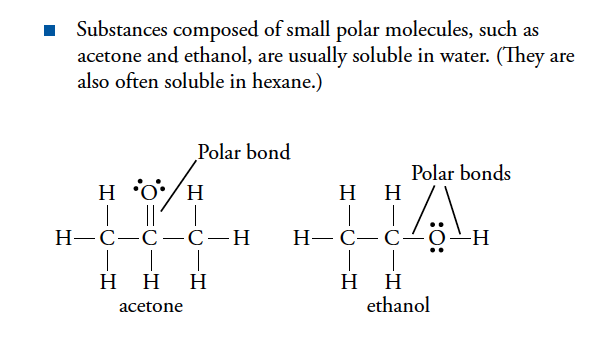
- Polar compound will dissolve in water
- compounds that will contain charges ( + or -)
- Not all things are soluble in water.
- Non-polar molecules will not dissolve in to polar solution.
- Example:
- Like dissolves like.
- Polar solvents dissolve polar solutes.
- Nonpolar solvents dissolve nonpolar solutes.
- Polar substances and nonpolar substances generally remain separate.
- The relative ability of an atom in a molecules to attract shared electron to itself.
- Every soluble substances will have a limit on how much solute will go into solution.
- When this happens, the solution is called saturated.
- If you had more solute after the saturation point then it will stay solid.
- Unsaturated solution: this type of solution occurs with a solution has not reached the limit of the solute.
- Qualitative terms: concentrated solution and dilute solution.
- Solution Composition: mass percent (weight percent) = mass of solute / mass of solution * 100