Introduction
- Since ancient times, humans have used chemical changes to their advantage.
- Two applications of chemistry that were used before 1000 B.C. are the process of ores to produce metals for ornaments and tools and the use of embalming fluids.
- By 400 B.C., Greeks proposed that all matter was composed of four fundamental substances: fire, earth, water, and air.
- The next 2000 years of chemical history were dominated by alchemy.
- alchemy: a precursor of chemistry and speculative philosophy practiced in the Middle Ages and the Renaissance
- Alchemy major focuses were discovering methods for transmuting baser metals into gold, finding a universal solvent and an elixir of life.
- Alchemy can be traced back to Ancient Egyptian and Ancient Chinese.
- Robert Boyle
- Born 1627 and died 1691.
- Help develop the idea of the element.
- A substance was an element unless it could be broken down into two or more simpler substances.
- Example: Air could not be an element as the Greek believes because it could be broken down to simpler parts.
- Flaws: he held to the idea that metals were not elements because he believed that metals could change due to alchemy.
Section 1: The Elements
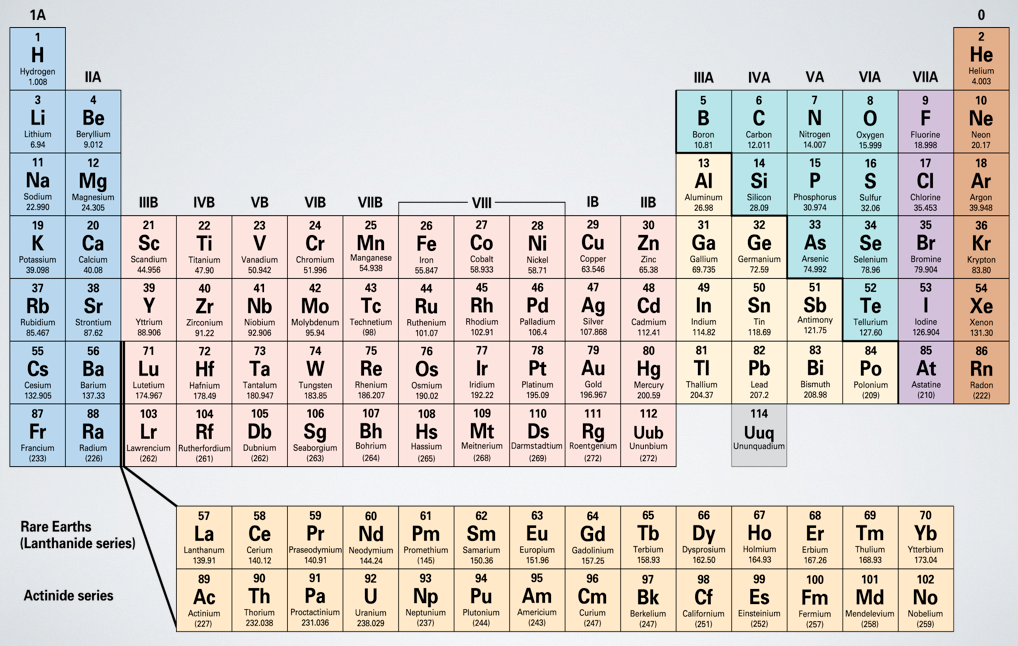
- Scientists have found that all matter can be broken down chemically into about 100 elements.
- However, about 116 different elements are known,* 88 of which occur naturally.
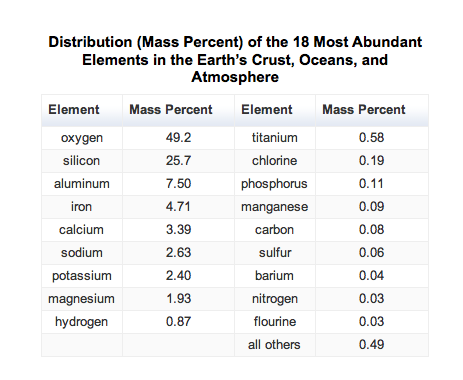
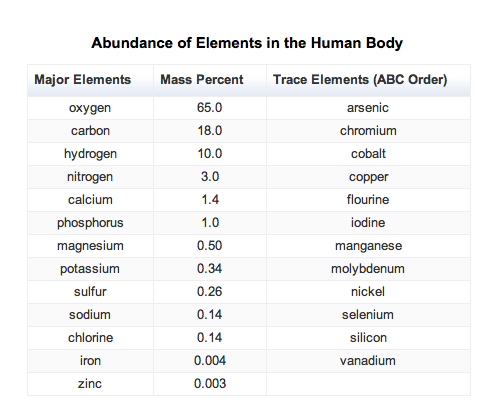
- Only 9 elements account for most of the compounds found in earth's crust.
Section 2: Symbols for the Elements
- Often an element's name is derived from a Greek, Latin, or German word that describes some property of the element.
- For example, gold was originally called arum, a Latin word meaning "shining dawn," and lead was known as plumbum, which means "heavy."
- Some of the heaviest elements are named after famous scientists-for example, einsteinium and nobelium.
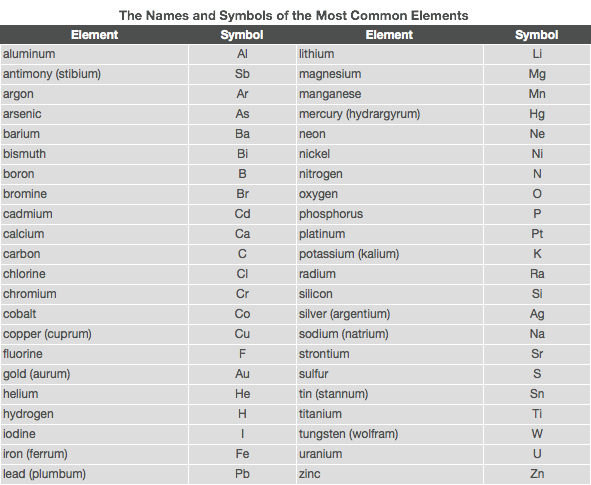
- Chemists have invented a set of abbreviations or element symbols for the chemical elements. These symbols consist of the first letter or the first two letters of the element names. The first letter is always capitalized, and the second is not.
- Examples: calcium = Ca nitrogen = N phosphorus = P
- Sometimes, the two letters used are not the first two letters in the name.
- Examples: strontium = Sr magnesium = Mg arsenic = As
- The symbols for some other elements are based on the original Latin/Greek name.
Section 3: Dalton's Atomic Theory
Section 4: Formulas of Compounds
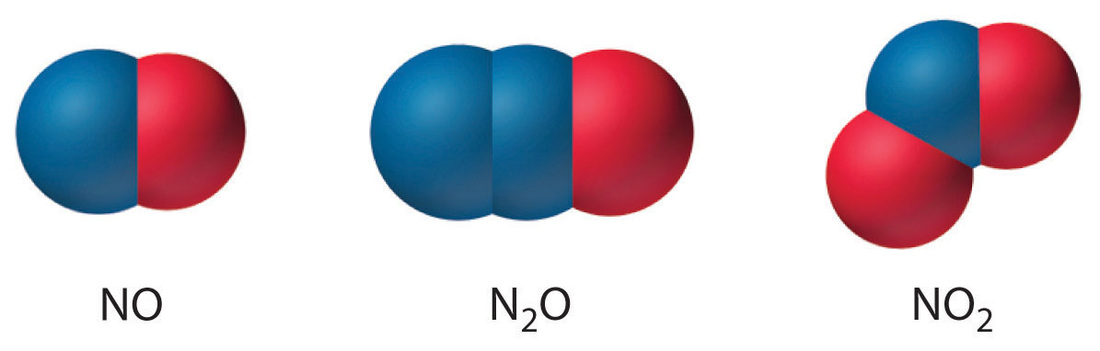
- The types of atoms and the number of each type in each unit (molecule) of a given compound are expressed by a chemical formula.
- In a chemical formula the atoms are indicated by element symbols, and the number of each type of atom is indicated by a subscript, a number that appears to the right of an below the symbol for the element.
- Example: The formula for water is written H₂O, showing that each molecule of water consists two atoms of hydrogen and one atom of oxygen.
Rules for Writing Formulas:
- Each atom present is represented by its element symbol.
- The number of each type of atom is indicated by a subscript written to the right of the element symbol.
- When only one atom of a given type is present, the subscript 1 is not written.
Example:
a. Each molecule of glucose, a type of sugar, contains six atoms of carbon, eleven atoms of hydrogen, and four atoms of oxygen.
Answer: C₆H₁₁O₄
Section 5: The Structure of the Atom
- Dalton’s atomic theory, proposed in the 1808, provided a convincing explanation for the composition of compounds that it was generally accepted.
- While people believed that atoms existed, they did not have an understanding on the structure of the atoms.
- A physicist, J.J. Thomson showed in the late 1890s that atoms of any element can be made to emit tiny negative particles.
- William Thomson believed that there most be a positive charge to balance the negative charges.
- William Thomson believed that there was a positive charge cloud with electron scattered to balance the charge.
Section 6: Introduction to the Modern Concept of Atomic Structure
|
Section 7: Isotopes
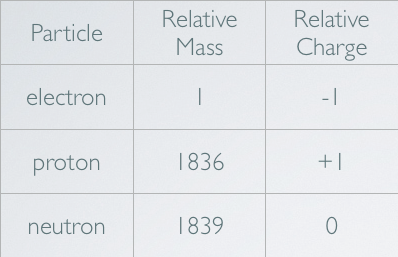
- For a given element, the number of neutrons will vary.
- The nucleus of the hydrogen atom has a single proton.
- A hydrogen atom with one neutron is called deuterium.
- A hydrogen atom with 2 neutrons is called tritium.
- The mass number of an element is approximately equal to number of protons plus the number of neutrons.
Section 8: Introduction to the Periodic Table
Section 9: Natural States of the Elements
- Most elements are reactive and combines to form new compounds.
- Some elements are not really reactive and can be found in the pure elemental states.
- Nobel: this term is given to elements that has low reactivity
- Nobel Metals:
- Gold, Silver, and Platinum
- Nobel Gases are all of the elements in group 8.
Section 10: Ions
- Ions are atoms that has either gain or loss one or more electron to form a charged atoms.
- Cation: atoms that has loss one or more electron to form a positive ion (Ca +2)
- Anion: atoms that has gain one or more electron to form a negative ion (O -2)
Section 11: Compounds That Contain Ions
- Chemical compounds contain ions.
- Whenever a compound forms between a metal and a nonmetal, it can be expected to contain ions.
- A chemical compound must have a net charge of zero.
- Both positive ions (cation) and negative ions (anions) must be present.
- The numbers of cations and anions must be such that the net charge is zero.
- How to write formulas for ionic compounds:
- Ca2+ and Cl- > CaCl2