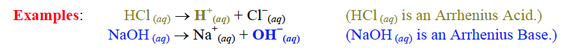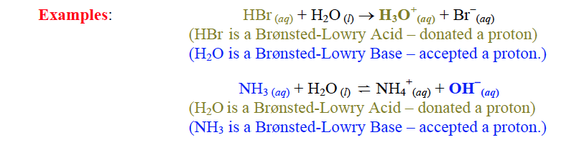Acids and Bases
Types of Acids/Bases
|
Strong VS. Weak Acid/Base
List of Strong Acids
|
|
conjugate base: the product formed after the acid donated a H+; behaves like a base when the reaction is looking from reverse
(acid > conjugate base) conjugate acid: the product formed after the base accepted a H+; behaves like a acid when the reaction is looking from reverse (base > conjugate acid) conjugate acid-base pair: the (acid/conjugate base) or (base/conjugate acid) pairs |
Ion-Product Constant
- Kw is called ion product constant of water.
- units are customarily omitted
- Kw = [H3O+][OH-]
- Remember that at 25 degree C.
- [H3O+] = [OH-] = 1.0x10-7M
- Kw = [1.0x10-7M][1.0x10-7M]
- Kw = 1.0x10-14
- The Kw of water will always be 1.0x10-14.
Ionization of Water
- Involves the transfer of a proton from one water molecule to another to produce a OH- and H3O+.
- In pure water only a small amount of OH- and H3O+ at 25 degree C.
- [H3O+]=[OH-]=1.0x10-7M
Understanding Logarithmic Function
- multiplication = short cut for addition
- exponential = short cut for multiplication
- logarithmic = short cut for exponential
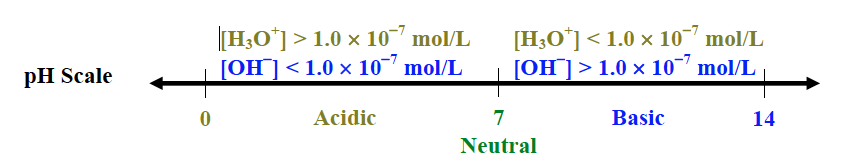
pH Scale
- a logarithmic scale to measure the acidity (relative [H3O+]) of a solution
- the lower the pH, the more acidic (less basic) is the solution (more [H3O+] and less [OH-])
- the higher the pH, the more basic is the solution (less [H3O+] and more [OH-])
- between 0 to 14, with 7 as neutral; but can be above 14 (very basic) or below 0 (very acidic)